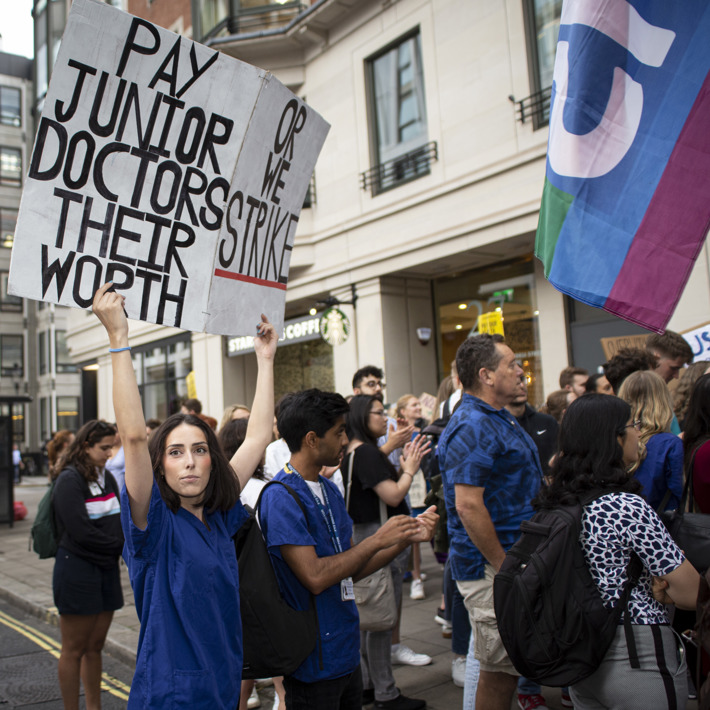Update your membership details
In order to properly represent the diversity within the medical workforce and student body, please update your equality data via your member profile when you join or log in.
The information you provide is kept strictly confidential and handled in accordance with the GDPR.
We fight for you

The Doctor
March 2024
Social insecurity
In the second of a two-part series, Peter Blackburn reports from the front line of the Deep End – this time shadowing a Glasgow GP inspired to advocate for the most disadvantaged in society
We give you a voice

Join our pay campaigns
- Consultants in England have accepted the latest Government offer on pay and DDRB reform.
- Consultants in Wales have suspended strike action in April to enter talks with Welsh government.
- Consultants in Northern Ireland ballot for industrial action will open on 7 May 2024.
- GPs in England members have overwhelmingly voted to reject the Government and NHS England’s 2024/25 GP contract changes.
- Junior doctors in England re-ballot to extend the mandate for industrial action, and provide us with a mandate for ASOS closed 20 March.
- Junior doctors in Wales committee has agreed to enter negotiations with the Welsh Government with a view of resolving the current pay dispute.
- Junior doctors in Northern Ireland voted in favour of industrial action and staged a full walk out in March.
- SAS doctors in England rejected a pay offer following a majority no vote and have re-entered negotiations with the Government.
- SAS doctors in Wales have suspended strike action in April to enter talks with Welsh government.
- SAS doctors in Northern Ireland voted in favour of taking industrial action in a indicative ballot.
You must be a BMA member to vote.
You must update your membership details to vote.
Get involved
Taking care of you
For questions about any aspect of your working life, our advisers are here to help you.
Opening times: 8am - 6pm Monday to Friday (excluding UK bank holidays)
What our members think
 The adviser I spoke to...was fantastic, incredibly well informed and really, really helpful. Your employment advisers are absolutely superb, thank you.Consultant
The adviser I spoke to...was fantastic, incredibly well informed and really, really helpful. Your employment advisers are absolutely superb, thank you.Consultant